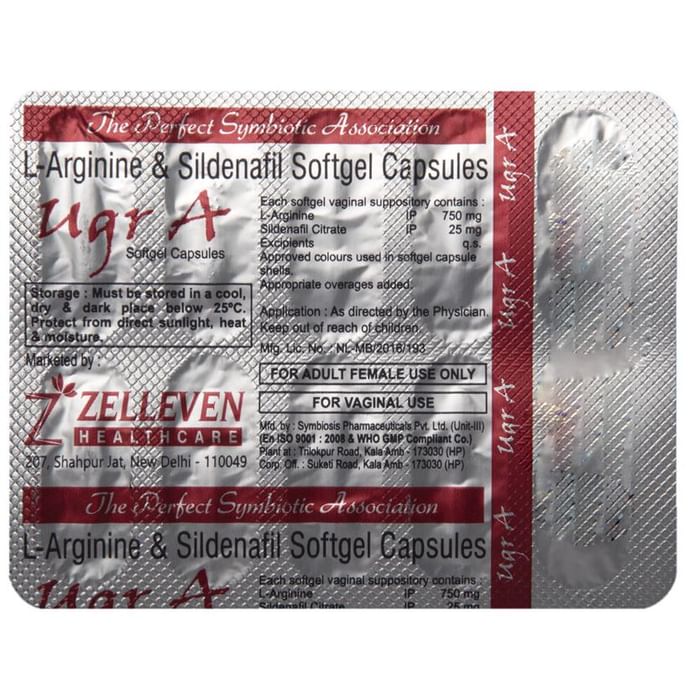
Product Informations: UGR A Softgel Capsule
Information About UGR A Softgel Capsule
In Intrauterine growth restriction (IUGR) Intrauterine growth restriction is a condition in which the growth of the fetus (unborn baby) slows or stops inside the mother’s womb during pregnancy. This is one of the most serious complications of pregnancy. UGR A Softgel Capsule treats this condition by increasing blood flow to the fetus. This helps provide nutrition and thus improve the birth weight of the fetus. UGR A Softgel Capsule gives a new hope to women whose pregnancies are complicated with IUGR. It is advisable to take UGR A Softgel Capsule as prescribed by the doctor.
Benefits
Description
How to use
Common Side Effect
- Diarrhea
- Headache
- Nosebleeds
- Pain in extremities
- Flushing (sense of warmth in the face, ears, neck and trunk)
Safety Advice
No interaction found/established
UGR A Softgel Capsule is generally considered safe to use during pregnancy. Animal studies have shown low or no adverse effects to the developing baby; however, there are limited human studies.
UGR A Softgel Capsule is probably safe to use during breastfeeding. Limited human data suggests that the drug does not represent any significant risk to the baby.
No interaction found/established
No interaction found/established
No interaction found/established
Missed dose
How Its Works
Fact Box
Question and Answer
Marketer details
FDC Ltd | B-8, MIDC Area, Waluj - 431 136, Dist. Aurangabad, Maharashtra
Alternatives and Substitutes
1 AL AX 5mg/75mg Capsule
Marketer/Manufacturer: Abbott || ₹14.5/tablet (25% cheaper)
Customer Reviews
No customer reviews yet.
References
FDA approved prescribing information. Levocitrizine; 1995 [revised May 2007]. [Accessed 01 Apr. 2019] (online) Available from: Read More
European Medicne Agency. Revised assessment report: Ambroxol and bromhexine containing medicinal products. 2015. [Accessed 01 Apr. 2019] (online) Available from: Read More
ScienceDirect. Ambroxol. [Accessed 01 Apr. 2019] (online) Available from: Read More
Levocitrizine. Slough, Berkshire: UCB Pharma Limited; 2007 [revised 27 Mar. 2019]. [Accessed 01 Apr. 2019] (online) Available from: Read More